- Blog
- Iar arm download
- How to add games to myboy emulator
- Urdu poem recitation
- What is mono framework
- Marine aquarium 3 beta
- Buku filsafat politik tahun 1989
- Xbox 360 fifa street 3 cheats
- Upgrade mac os x 10-6 8 to 10-7
- Is euro truck simulator 2 multiplayer
- Ros matlab simulink
- A2 carry handle upper
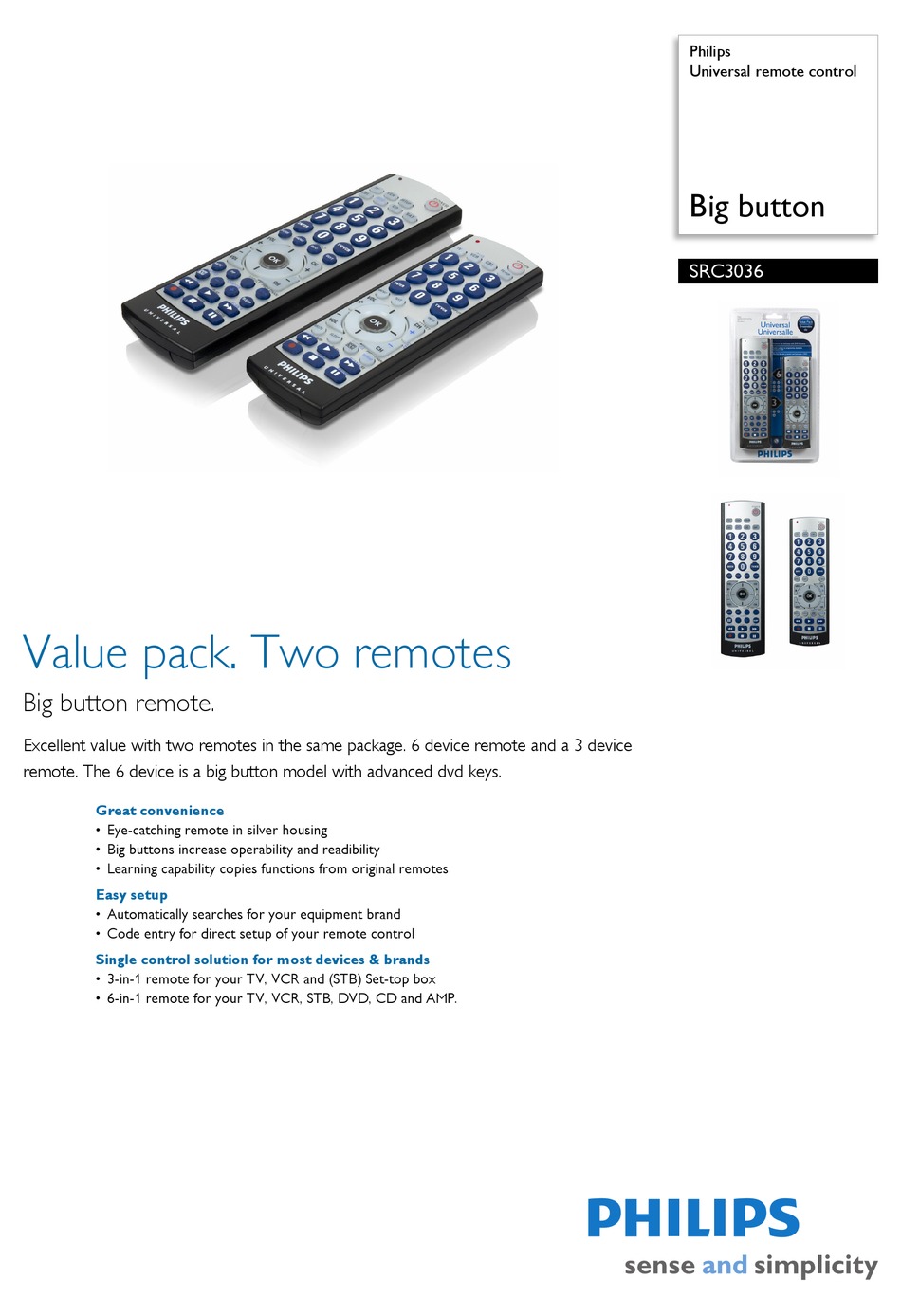
- Www philips com src update
- Blog
- Iar arm download
- How to add games to myboy emulator
- Urdu poem recitation
- What is mono framework
- Marine aquarium 3 beta
- Buku filsafat politik tahun 1989
- Xbox 360 fifa street 3 cheats
- Upgrade mac os x 10-6 8 to 10-7
- Is euro truck simulator 2 multiplayer
- Ros matlab simulink
- A2 carry handle upper
- Www philips com src update
- #WWW PHILIPS COM SRC UPDATE SERIAL NUMBER#
- #WWW PHILIPS COM SRC UPDATE PDF#
- #WWW PHILIPS COM SRC UPDATE REGISTRATION#
- #WWW PHILIPS COM SRC UPDATE FREE#
We are committed to resolving this issue and providing transparent, ongoing communication as we navigate the next steps. We are committed to holding ourselves to the highest standards of product quality and safety in an effort to do what is right for you, and the patients who trust you with their care. We recognise the importance of providing safe and effective therapy.
#WWW PHILIPS COM SRC UPDATE PDF#
Please see the FAQs below and Clinical Information PDF here.įor the past 40 years we have centered our business around our commitment to patient care, with solutions that are aimed at improving the lives of people with respiratory and sleep challenges. To implement the permanent corrective action, where possible, Philips will be contacting affected patients and customers with the relevant information on the next steps. With this, your patients are likely to contact you for guidance and we are committed to providing you with information and tools to have an informed discussion with them and make your clinical recommendation. The Urgent Product Defect Correction informs customers and users of potential impacts on patient health and clinical use related to this issue, as well as instructions on actions to be taken. The foam degradation may be exacerbated by use of unapproved cleaning methods, such as ozone (see FDA safety communication on use of ozone cleaners ), and off-gassing may occur during initial operation and may possibly continue throughout the device’s useful life.įor more information on this Urgent Product Defect Correction, including a list of affected devices, please visit our main Urgent Product Defect Correction page.
#WWW PHILIPS COM SRC UPDATE FREE#

You can access the Philips Respironics Q&A at.Please also reach out to your physician with any additional questions you may have regarding your therapy.At this point, Philips has not provided the timing of the availability of potential future product replacement.Medic Home Health Care is in communication with Philips about this recall and we will do our best to communicate information to patients as it becomes available.

For information on the Recall Notice, a complete list of impacted products, and potential health risks, visit On June 14, 2021, Philips Respironics announced a voluntary recall for Continuous and Non-Continuous Ventilators (certain CPAP, BiPAP, and Ventilator Devices) due to two issues related to the polyester-based polyurethane (PE-PUR) sound abatement foam used in these devices.Please be sure to read this information in detail. We want to make you aware of an important development related to certain CPAPs, BiPAPs, and ventilators manufactured by Philips Respironics.
#WWW PHILIPS COM SRC UPDATE REGISTRATION#
More countries will be added shortly.ĬLICK HERE TO BEGIN THE REGISTRATION PROCESS At this time, this process is only available for patients, users, or caregivers in the USA and Canada.
#WWW PHILIPS COM SRC UPDATE SERIAL NUMBER#
Philips has established a registration process that allows patients, users, or caregivers to look up their device serial number and begin a claim if their unit is affected.
